- Blog
- Princess maker 4 english patch download
- Booka shade movements 10 beatport
- Nxpowerlite desktop 6 portable
- Lightroom 6-2 1
- Insanity max 30 sweat intervals rutube
- Pcsx2 pc
- Soch hardy sandhu karaoke
- Homage inverter troubleshooting
- Wpap tutorial
- Avisynth media frame server
- Descargar mac os mojave dmg
- Aria made in japan model 8715 12 string
- Bio-rad cfx manager 3-1 add freeze forever step
- Watch cardcaptor sakura the movie 1 english subbed
- Day autocad 2007
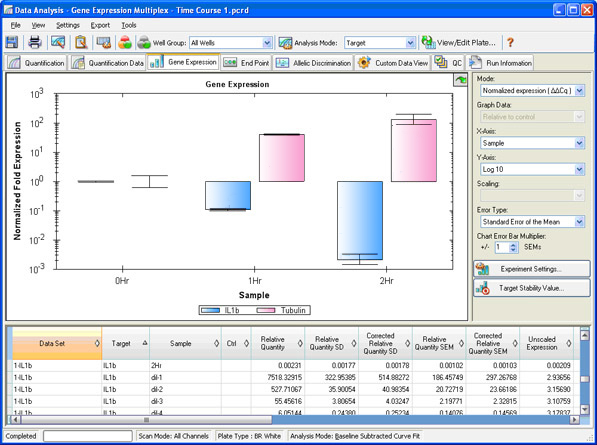
CYP6P3 and CYP6M2 are considered as the main pyrethroid metabolising enzymes in several An. Several cytochrome P450-dependent monooxygenases have been functionally associated with pyrethroid resistance. In Anopheles mosquitoes, resistance is primarily conferred by mutations at the insecticide’s target site that alter its sensitivity, and by the upregulation of enzymes that detoxify or sequester the insecticide. The resulting insecticide resistance (IR) in the major malaria vector Anopheles gambiae ( s.l.) represents one of the greatest challenges in malaria control. However, the increasing use of a limited number of insecticides, primarily pyrethroids, places an immense selection pressure on insect populations, which has not left disease vectors unaffected. Insecticide based vector control interventions have reduced malaria incidence. They may complement current diagnostic assays to provide evidence-based and operationally relevant information for insecticide resistance management. gambiae ( s.l.) are simple to perform, robust and rapid. The novel multiplex assays for monitoring the levels of major detoxification genes and metabolic resistance in An. The assays can also be applied directly on lysates of mosquito specimens, without RNA extraction or DNase treatment. The assays were successfully validated in terms of expression levels against standard two-step singleplex qPCR assays (overall % difference = -17.6%, 95% CI = -38.7-3.43%) and microarrays, using laboratory strains and field-caught samples. The novel assays are efficient (reaction efficiencies = 95-109%), sensitive (covering a > 10.0 Ct range with R 2 values > 0.99), specific (TaqMan chemistry), reproducible (%CV = 4.46–12.07%), as well as readily expandable to capture additional loci as they evolve or to cover additional species. The assays were also tested directly on lysates of RNA later®-preserved mosquitoes without an RNA extraction step. The novel assays were validated versus gold standard methods with a range of phenotyped mosquito specimens. Primers and probes were designed to target the mRNA of cytochrome P450-dependent monooxygenases CYP6P3, CYP6M2, CYP9K1, CYP6P4 and CYP6Z1, and the glutathione-S-transferase GSTE2. Rapid one-step multiplex TaqMan-probe based RT-qPCR assays were developed and optimised to measure the expression levels of genes associated with metabolic insecticide resistance in An. However, detection of metabolic resistance is not trivial and relies on laborious bioassays, unspecific biochemical methods, or sophisticated and expensive molecular approaches using transcriptomics. Metabolic resistance of the major malaria vector Anopheles gambiae ( s.l.) to insecticides is operationally significant, particularly in combination with target site resistance.